
Acclivity Technologies is a discovery to preclinical health technologies company based in North Carolina.
We are assessing the measurability of ATI-0019, a novel small molecule capable of modulating endothelial cell activity, for use as a biomarker in research, consumer health, wellness and aging applications and clinical diagnostics, as well as assessing the pharmacological properties of ATI-0019 and developing a library of related compounds for therapeutic use in aging related cardiovascular and other diseases. Future investigations will explore potential drug/device combinations, smart devices, and diagnostic applications.
Rooted in the realms of biochemistry and endothelial cell biology, and fortified by years of experience in R&D leadership and product development, ATI is driven by the collective expertise of its founders. With a vision that transcends conventional boundaries, we are poised to redefine consumer health, wellness, and aging solutions through pioneering research and strategic leadership. Our founders' profound understanding of biochemical intricacies and endothelial cell dynamics forms the cornerstone of our approach. Strategic foresight and unwavering commitment to excellence propel us toward groundbreaking advancements, as we strive to address unmet medical needs with precision and dedication. Guided by our vision, ATI stands at the forefront of innovation, poised to empower individuals worldwide to take control of their health, embrace vitality, and thrive at every stage of life.
Features of ATI’s integrated product development approach include:
- Biomarker-Driven Health Assessment Tools: ATI will leverage our research expertise to identify and validate biomarkers associated with health, wellness, and aging. These biomarkers will then be integrated into consumer health assessment tools, providing personalized insights into an individual's health status and aging-related risks. Consumers will be able to track their biomarker profiles over time to monitor changes and make informed decisions about their health and wellness strategies.
- Diagnostics Integration: ATI will further advance our offerings by integrating biomarkers into diagnostic tools for early detection of age-related diseases and health conditions. By combining biomarker data with other health metrics, such as genetic predispositions and lifestyle factors, these diagnostics will provide comprehensive risk assessments and facilitate early intervention strategies.
- Drug Development: Building upon the identification of biomarkers, ATI will prioritize the development of small molecule drugs targeting specific biomarker pathways associated with age-related diseases and health conditions. Utilizing its lead drug candidate and endogenous biomarker, ATI-0019, as a blueprint, ATI will design and optimize small molecule drugs to modulate biomarker activity and mitigate disease progression in conditions such as cardiovascular diseases, inflammation, and age-related disorders.
- Personalized Therapeutic Interventions: By incorporating biomarker data into treatment algorithms, ATI will offer personalized therapeutic interventions tailored to an individual's biomarker profile and health needs. This personalized approach will optimize treatment efficacy, minimize adverse effects, and improve overall health outcomes for consumers.
- Clinical Validation and Regulatory Approval: ATI will conduct clinical studies to validate the utility of biomarkers for diagnostics and treatment response prediction. These studies will provide the necessary evidence to support regulatory approval of biomarker-driven diagnostics and small molecule drugs, paving the way for their widespread adoption in clinical practice and consumer healthcare settings.
- Integrated Health Platforms: ATI will integrate biomarker-driven health assessment tools, diagnostics, and therapeutic interventions into comprehensive health management platforms for consumers. These platforms will offer seamless integration of biomarker data, personalized health recommendations, remote monitoring capabilities, and access to ATI's portfolio of small molecule drugs for targeted intervention.
By integrating biomarker development into its consumer health, wellness, and aging initiatives, ATI will offer innovative solutions that empower individuals to proactively manage their health, detect age-related diseases at an early stage, and receive personalized treatment interventions tailored to their unique biomarker profiles.
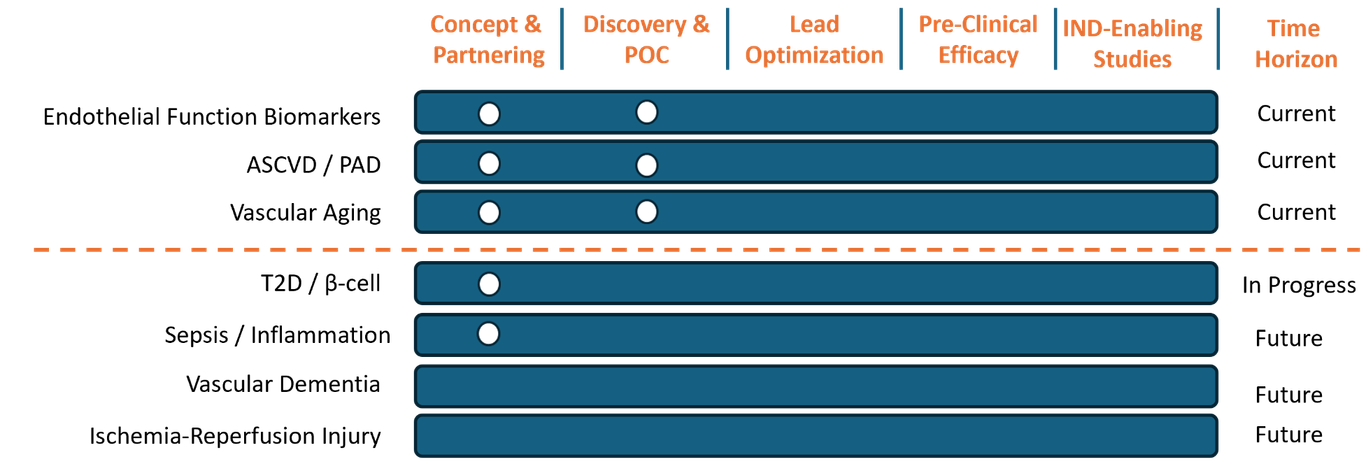
Acclivity Technologies Inc. Pipeline
© Copyright. All rights reserved.
We need your consent to load the translations
We use a third-party service to translate the website content that may collect data about your activity. Please review the details and accept the service to view the translations.